New software designs optimized, personalized treatments for movement impairments
First-of-its-kind computer-aided engineering tool could aid clinical treatment design.

B.J. Fregly is a professor of mechanical engineering and bioengineering, a CPRIT Scholar in Cancer Research, and a recent Brown College magister at Rice University. Photo by Jeff Fitlow/Rice University
Silvia Cernea Clark, Rice University July 27, 2023
A team of Rice University engineers has launched a first-of-its-kind, open-source software that constructs and uses personalized computer models of how individual patients move to optimize treatments for neurologic and orthopedic mobility impairments.
Funded by a grant from the National Institutes of Health, the Neuromusculoskeletal Modeling (NMSM) Pipeline software developed by B.J. Fregly and collaborators in the Rice Computational Neuromechanics Lab is now available to clinician/engineer teams that would like to utilize computer-aided engineering for clinical treatment design.
The software could be used to design orthopedic surgical plans, neurorehabilitation interventions, physical therapy regimens and prosthetic devices that maximize recovery of lost function for patients with movement impairments caused by stroke, osteoarthritis, cerebral palsy, Parkinson’s disease, spinal cord injury, traumatic brain injury, limb amputation and even some forms of cancer.
“Computer-aided engineering (CAE) software revolutionized the design of airplanes, automobiles and industrial equipment in the 1990s,” said Fregly, a professor of mechanical engineering and bioengineering and CPRIT Scholar in Cancer Research. “When I worked as a CAE software developer in industry during that time period, I kept thinking, someone needs to do this for the design of treatments for movement impairments.”
Many years in the making, the NMSM Pipeline builds upon existing software used to model, simulate and analyze the musculoskeletal system by making it useful for clinical treatment design.
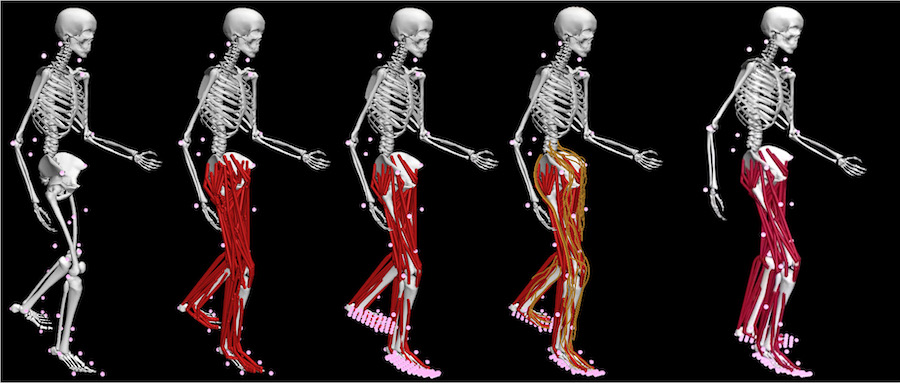
Schematic of the four steps involved in the model personalization process, from personalizing joint models, to muscle-tendon models, to ground contact models, to neural control models, resulting in a final personalized model that can be used to predict the patient’s posttreatment walking function. Courtesy of the Fregly group/Rice University
“Our software makes it easy to create a personalized computer model of a patient’s neuromusculoskeletal system using the patient’s pretreatment movement data, and it then uses that model to predict ⎯ and even optimize ⎯ the patient’s functional outcome for various treatment designs that a clinician wants to explore,” Fregly said. “For clinical utility, you need these two things ⎯ model personalization and treatment optimization.
“Instead of relying on implicit, subjective predictions of a patient’s post-treatment function, clinicians working with engineers could use our software to make explicit, objective predictions, which could not only weed out ineffective or harmful treatments, but also generate highly effective unforeseen ones.”
The physics-based software integrates several different physiological models, including models of central nervous system (CNS) control, muscle force generation and metabolic energy expenditure.
“The model of how the CNS coordinates muscle activity is based on analysis of electromyographic (muscle electrical activity) data collected during human movement,” Fregly said. “Rather than controlling a large number of muscles individually, the CNS appears to control a small number of coordinated muscle groups called ‘synergies.’

Rice Computational Neuromechanics Lab members Geng Li (from left), B.J. Fregly, Kayla Pariser, Spencer Williams, and Marleny Vega. Photo by Jeff Fitlow/Rice University
“What’s really interesting is that if you analyze electromyographic data, you’ll find that ⎯ compared to healthy individuals ⎯ people who’ve had a stroke tend to have fewer synergies available to construct their muscle activations,” Fregly said. “You might say they’ve lost one or two neural control building blocks as a result of the stroke, which means they can’t generate the same repertoire of signals to control their muscles, and so they can’t move as well.
“One of the challenges for stroke rehabilitation is, can you find ways to train people’s brains so that you can help them recover lost synergies? We think we can predict how to do that. We’re not there yet, but we’re close.”
Using data from an individual post-stroke walking at their self-selected speed of 0.5 meters per second, Fregly’s group generated three different predictions of how the subject would walk at their fastest-comfortable speed of 0.8 meters per second.
“One prediction assumed we had torque actuators, like in a robot, controlling the lower body joints, so no muscles,” Fregly said. “A second prediction assumed we had muscle actuators, but all muscles were controlled independently. The third prediction also assumed we had muscle actuators, but now all muscles were controlled using a small number of synergies.

Claire Hammond is a mechanical engineering doctoral student in the Computational Neuromechanics Lab at Rice University. Photo courtesy of Claire Hammond
“As our model moved from torque controls to individual muscle controls to synergy controls, it became more realistic as well as personalized physiologically. Interestingly, the approach that was the most physiological ⎯ using synergy controls ⎯ also produced the most accurate prediction of how the subject walked at their fastest comfortable speed. It was like magic.”
Pelvic cancer surgery is another clinical example where the NMSM Pipeline could help guide treatment decisions.
“You can’t cookie-cutter surgical treatments for people who get pelvic cancer,” Fregly said. “Oftentimes, a patient will lose a hip joint. For some patients, the best surgical option might be to implant a custom prosthesis that replaces lost bone and hip joint function, while other patients might benefit more from a surgical option that does not replace the removed bone and hip joint.
“For both options, we are already collecting pre-surgery walking data from individual patients, creating a personalized computer model of each patient, and predicting each patient’s post-surgery walking function given knowledge of the implemented surgical decisions. Once we can closely match each patient’s post-surgery walking data, we will be ready to inform actual surgical planning with the goal of maximizing each patient’s post-surgery walking function.”

Di Ao is an assistant professor and postdoctoral associate at the Institute of Medical Research at Northwestern Polytechnical University, China. Photo courtesy of Di Ao
A computational simulation tool called HeartFlow is already being used clinically to determine optimal stent placement for patients who undergo cardiac procedures.
“HeartFlow combines a scan of a patient’s relevant anatomy with a computational fluid dynamics model that simulates blood flow through the arteries in the heart,” Fregly said. “Doctors can use it to test different stent sizes and placements to determine what would work best for the patient. We hope that the NMSM Pipeline will eventually be used in a similar manner to inform treatment design for movement impairments.”
To encourage the research community to use the software, Fregly and his team are running a four-year competition at the American Society of Biomechanics annual conference. The competition will challenge researchers to develop personalized treatments that improve walking function for four individuals who have had a stroke.
“Each year, we’re going to provide researchers with extensive experimental walking data from a different individual post-stroke so that they can construct a personalized model of each individual and use it to design a personalized treatment computationally,” Fregly said. “They could try strengthening specific muscles, or knocking out specific muscles with botox, or modifying neural control strategy, or training with a split-belt treadmill or rehabilitation robot, or designing an exoskeleton ⎯ whatever treatment they want to explore, they can simulate it. The goal is to enable the individual being modeled to walk at a normal speed with a normal motion.”
![]() Source Rice University
Source Rice University
Also see
NIH funds effort to customize treatment of movement impairments Rice University
